Chemistry (Triple)
Structures, Trends, Chemical Reactions and Analysis
Elements
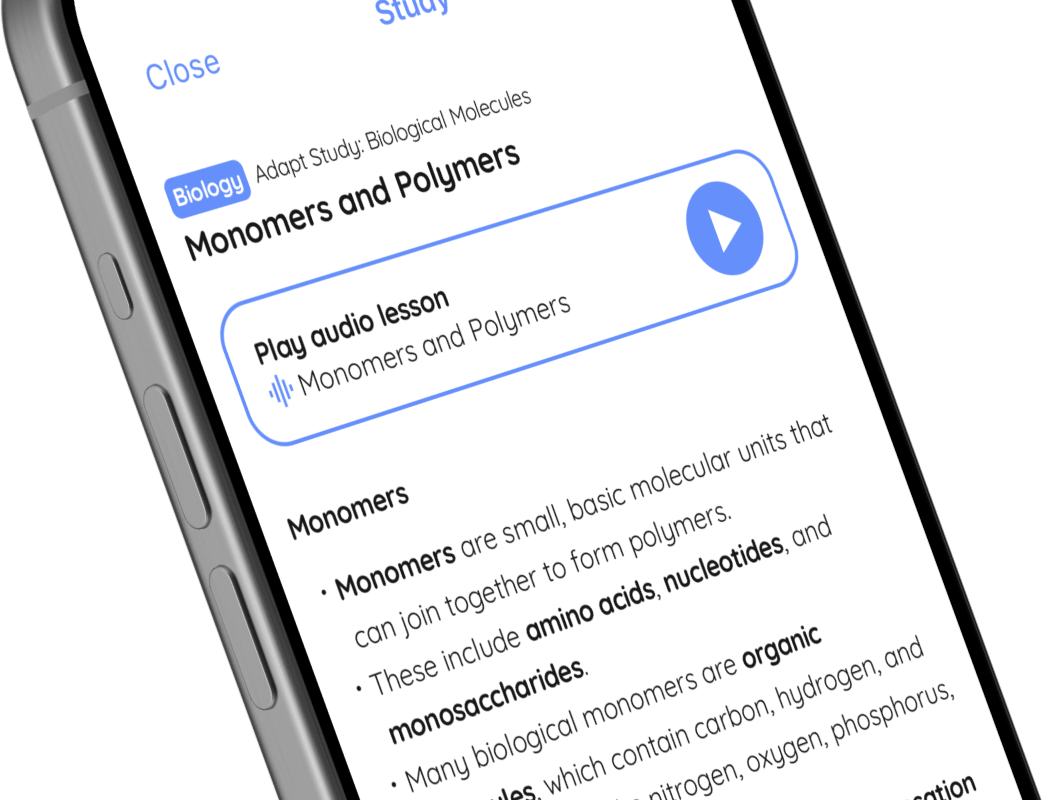
🤓 Study
📖 Quiz
Play audio lesson
Elements
Understanding Elements
- Elements are substances that consist of just one type of atom.
- Each element is represented by a unique chemical symbol, typically one or two letters e.g., H for Hydrogen, O for Oxygen, Mg for Magnesium.
- Elements can be metallic (metals), non-metallic (non-metals), or metalloids (possess properties of both metals and non-metals).
Arrangement in Periodic Table
- The periodic table organises elements based on their atomic number and chemical properties.
- Element's position in the periodic table determines its group number (vertical columns) and period number (horizontal rows).
- Elements in the same group usually have similar chemical properties because they have the same number of electrons in their outer shell.
- Metallic elements are on the left side of the periodic table, while non-metallic elements are on the right.
Atom Structure
- Atoms of an element consist of a nucleus, containing protons and neutrons, with electrons orbiting around the nucleus.
- Protons are positively charged, electrons are negatively charged and neutrons have no charge.
- The number of protons in an atom gives the atomic number of the element, and the sum of protons and neutrons gives the mass number.
Chemical and Physical Properties
- Chemical properties of an element describe its behaviour in chemical reactions, including reactivity and valency.
- Physical properties include melting and boiling point, density, and conductivity of heat and electricity.
- Within a group in the periodic table, the reactivity of metals increases down the group while the reactivity of non-metals decreases down the group.
Isotopes
- Isotopes are atoms of the same element that have different numbers of neutrons and consequently, different mass numbers.
- Isotopes of an element have identical chemical properties but can differ in physical properties such as density and rate of diffusion.
Elemental Analysis
- Elemental analysis can be conducted using techniques such as spectroscopy, mass spectrometry, and chemical tests.
- These techniques can determine the identity of an element, its concentration, or its atomic structure.