Applied Science
Fundamentals of Science
Practical and scientific skills
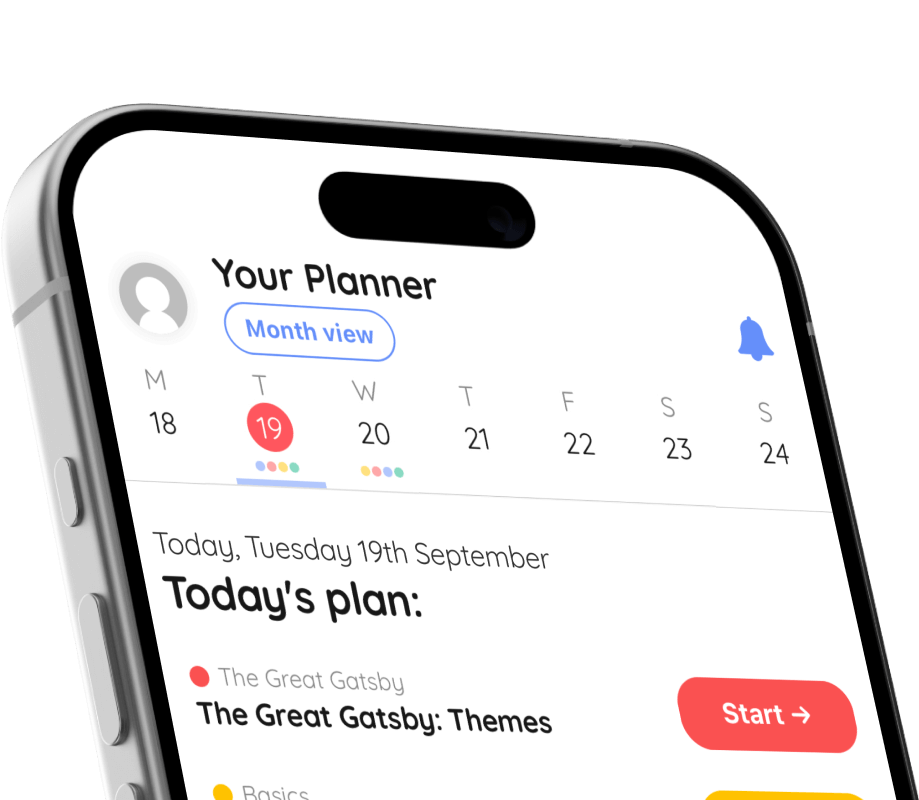
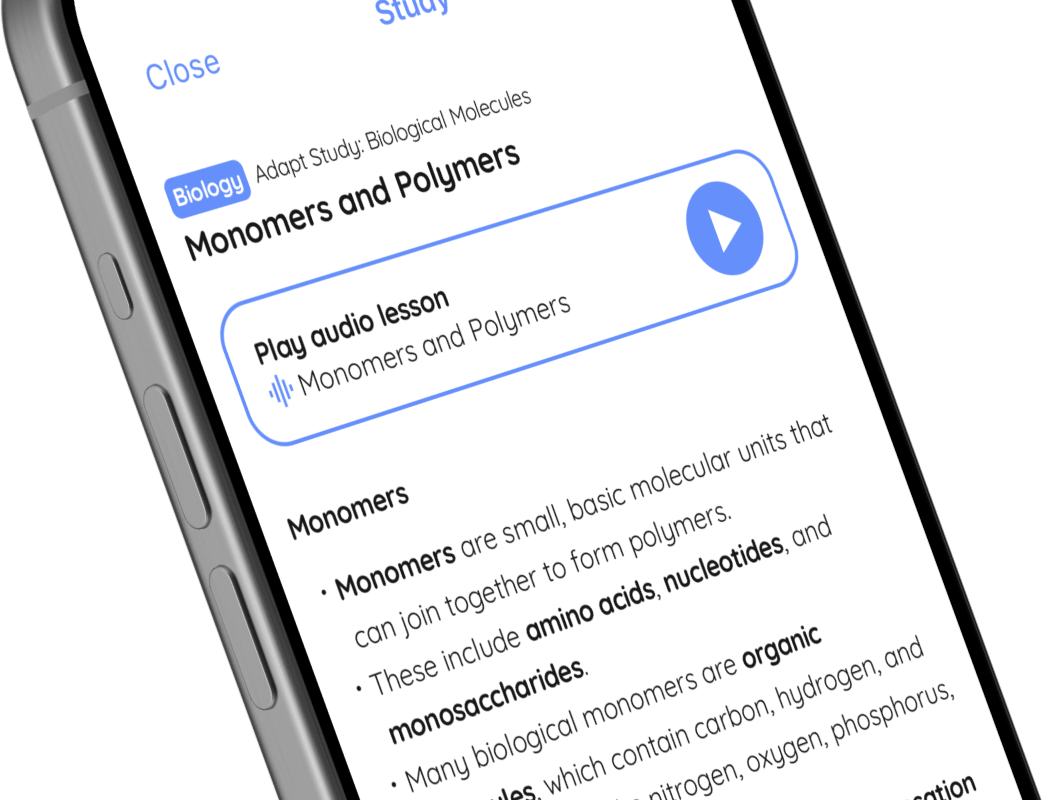
🤓 Study
📖 Quiz
Play audio lesson
Practical and scientific skills
Section 1: Understanding and Planning Scientific Investigations
- Formulating a precise scientific question is the basis of a scientific investigation.
- Propose a hypothesis that aligns with the scientific question and consider potential predictions.
- Carefully plan experiments to test the predictions based on the hypothesis, taking into account independent, dependent and controlled variables.
Section 2: Experimental and Practical Skills
- Precision and accuracy are key in all scientific measurements. Use appropriate equipment to ensure accurate data collection.
- Always follow health and safety procedures while conducting experiments. Understand the hazards associated with materials and equipment.
- Replicate experiments to confirm findings and minimise anomalies.
- Develop the ability to record observations systematically during an experiment.
Section 3: Data Analysis
- Approach data with a rigorous and critical mindset. Discard anomalous results that may skew the final analysis.
- Utilise appropriate statistical methods to analyse data. Find patterns and correlations and understand their significance.
- Understand the limitations of collected data and any uncertainties within it.
Section 4: Evaluation and Communication
- Evaluate the quality of the experiment. Consider methodical limitations and how they may affect the validity of the result.
- Generate conclusions based on the data analysis. Ensure they substantiate the hypothesis.
- Communicate findings effectively. Use diagrams, graphs, or charts to visualise data and write concise, clear explanations.
- Devise follow-up experiments based on findings, indicating a logical progression in scientific understanding.
Section 5: Ethical, Social, and Environmental Considerations
- Understand the ethical considerations relating to scientific work, e.g. clinical trials, animal testing, and data privacy.
- Consider the social and environmental impact of scientific investigations and outcomes.
- Develop a sense of responsibility, integrity, and care for sustainability and biodiversity, adhering to scientific and environmental ethics.