Physics (Combined)
Physics
Forces and their Interactions
🤓 Study
📖 Quiz
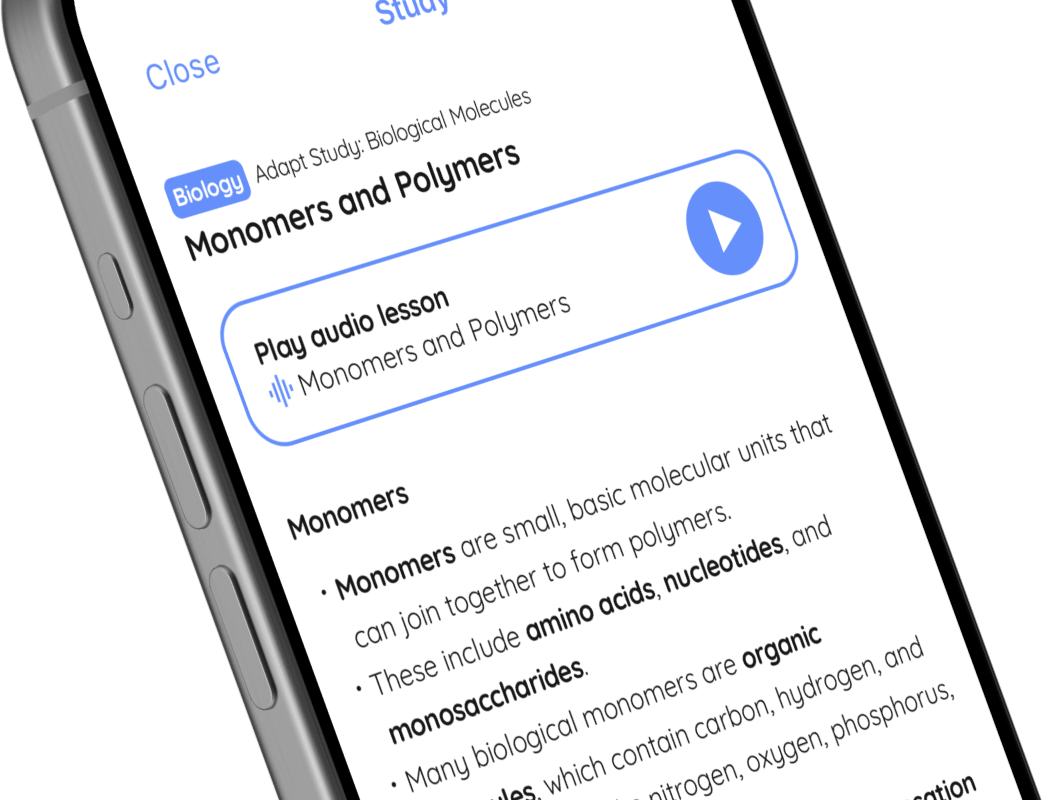
Play audio lesson
Forces and their Interactions
Vector and Scalar Quantities
- Scalar quantities are entirely described by a magnitude (size) only.
- Examples of scalar quantities include speed, distance, mass, temperature and energy.
- Vector quantities have both a magnitude and a direction.
- Examples of vector quantities are force, velocity, displacement, acceleration and momentum.
Newton's First Law of Motion
- The first law of motion states that an object will remain at rest or continue in a straight line at a constant speed unless acted upon by an external force.
- This property of resistance to change in motion is known as inertia.
Newton's Second Law of Motion
- According to Newton's second law of motion, force is the product of mass and acceleration. It's expressed as F = ma.
- The acceleration of an object is directly proportional to the net force acting on it and inversely proportional to its mass.
- The unit of force in the International System of units is the Newton (N) which is defined as 1 kg·m/s².
Newton's Third Law of Motion
- Newton's third law motion states that for every action, there is an equal and opposite reaction.
- This means that any force exerted onto a body will create a force of equal magnitude but in the opposite direction on the object that exerted the first force.
Principles of Forces
- Friction is a force that opposes motion. It occurs when two surfaces are in contact with each other.
- Air resistance is a type of friction that acts on objects moving through the air.
- The force of gravity near the Earth's surface is referred to as weight and is equal to an object's mass multiplied by the acceleration due to gravity.
- Centripetal force is the force that keeps a body moving with a uniform speed along a circular path and it's directed along the radius towards the centre.
- The equilibrium of forces occurs when the sum of forces acting on a body is zero, resulting in no change in motion.
Momentum
- Momentum is a vector quantity defined as the product of an object's mass and velocity.
- The principle of conservation of momentum states that the total momentum before and after a closed, isolated system interaction remains the same.